Significantly
Improved Mileage:
This article about acetone (CH3COCH3)
probably draws conclusions that Big Oil
and the American Car Manufacturers and
others do NOT want you to know. They suffer from unlimited
corporate GREED. They want bad mileage. The worse, the better as
far as they are concerned. Acetone is a vaporization additive
rather than a fuel additive per se. It is successful in
very tiny amounts from about one part per 6000 to one part per
2000. However above one part per 1000, the mileage seems to taper
off while HC emissions actually are greatly reduced. The peak gain
in mileage comes with between .075 of one-percent
and .20 of one-percent acetone, depending on the
actual vehicle which may be running gasoline or diesel. Acetone
operates on the unburned portion of the fuel to gain added
vaporization and improve combustion efficiency. There are no bad
effects and every good reason to use acetone in your fuel.
So try a little acetone.
Acetone is the secret additive for mileage. Containers labeled acetone
from a hardware store are usually okay and pure
enough to put in your fuel. We prefer cans or bottles that say
100-percent pure. The acetone in gallons or pints we get from
Fleet Farm are labeled 100% pure. The bottles from Walgreen say
100% pure. Never use solvents such as paint thinners or unknown
stuff in your gas. Toluene, benzene and xylene are okay if they
are pure but may not raise mileage except when mixed with acetone.
The author has used ACETONE in gasoline and diesel fuel and in jet
fuel (JP-4) for 50 years. He has tested fuels independently and is
an authority on this important subject. For instance a tiny bit of
acetone in diesel fuel can stop the black smoke when the rack is
all the way at full throttle. We recently proved that Carb Medic
from Gunk can raise mileage when 5 oz. are used with 2 oz. of
acetone per 10 gallons of gasoline, even in cold weather. Normally
arctic air prevents accurate mileage testing in the winter. And never
allow skin contact with these additives. Nor should you breathe
this stuff. Read the directions on the can.
Questions asked of
someone in the petroleum industry regarding ACETONE will
automatically trigger a string of negative reactions and perhaps
false assertions. We may have heard them all. The mere mention of
this additive represents such a threat to oil profits that you may
get fabricated denials against the successful use of acetone in
fuels. For this reason, test the stuff for yourself. The author
has never found any valid reason for not using acetone in gasoline
or diesel fuel. Plus it takes such a tiny amount to work. No
wonder they fear this additive. Of course you might Email this
article to your congressman because clearly ACETONE should be
ordered by Federal Law to be present in all fuels.
There is a great little
device available to check your exact gas mileage and more. See
ScanGauge.com for a very timely instrument
that fits any car 1996 or newer. See your real-time MPG, inlet
temperature and many more details as you drive. This inexpensive
tool should end a lot of debate over what works for mileage and
what does not.
There are of course
other additives that improve mileage but these have been
black-balled by the petroleum industry. The industry could easily
add these ingredients into gasoline and diesel fuel. But will they
dare to improve your mileage? NO. You just have to stumble onto
the fuel mileage secrets all by yourself, like we have. Certain
octane improvers for example also aid mileage. But unfortunately
many products claiming to improve mileage are expensive and do not
really help much. Others are fakes. For instance, a smooth flow of
air into a carburetor or injector is far better for mileage than
turbulent air. Yet many people deliberately introduce turbulent
air into their engines. There are many silly myths floating around
the car industry to fool the average person. Another is that cold
intake air improves mileage. NO. Warm
air improves mileage.
Test for yourself. Take
a mileage check for each and every tank of gas or diesel fuel like
we do. Your actual mileage is NOT that of a single tankful but the
average of perhaps five tanks worth. To be accurate, you should
not miss any checks. This takes discipline to get reliable
results. Someday your car will do it for you with an MPG gauge on
the dash. But for now, YOU ought to keep tabs on your mileage for
all our sakes. The ideal auto would save the MPG of your last tank
of gas on your instruments. Be consistent where you buy your
gasoline because different gasolines vary tremendously. The best
gas and the worst gas in your neighborhood will likely have a
30-percent spread in mileage, according to the author's
experience. Same for diesel fuel. Try to keep down the number of
variables wherever you gas up by using the same station, same
pump, same grade or same octane before testing. In almost all
cases, the lowest octane is best for mileage. Most modern vehicles
do not have high enough compression to justify using high octane
fuels. The testing indicates best mileage is obtained with 85 or
87 octane gasoline. Too much octane causes a loss of power and
economy. BUT too little octane causes the same things plus
knocking. Listen carefully to your engine for tell-tale knocks or
clicks when you start out from a light. Best mileage points to the
correct octane when the engine is properly tuned. Pure acetone was
rated in 1920 by Sir Harry Ricardo at 150 octane. Plus acetone
turned out to be an excellent additive to reduce exhaust emissions
in both gas and diesel engines.
The question we most
hear is, "Will it harm my engine?" We feel like saying,
"YES, it will blow it up." But instead we patiently
explain how over five decades, we have never seen a problem with
acetone. Would anyone use something more than once if it were
harmful? Not me. Think about that. Only an oil company stooge
would say anything negative about this nice additive.
Pure acetone or
propanone is an extremely clean burning fuel that burns in air
with a pretty blue smokeless flame. Acetone is a highly flammable
liquid. It also removes paint. Do not get it on your paint and do
not take it near a flame or spark. Acetone can reduce hydrocarbon
emissions up to 60-percent. In some older cars, the HC readings
with acetone went from say 440 PPM to 195, as just one example.
Acetone is toxic to breathe and should be stored outside, not
inside your house. Gasoline by itself is also highly toxic, so
treat them both with respect and great care. Just a precaution.
The author has soaked carburetor parts in acetone for months and
even years to see if there is any deterioration. None. Gasoline
and/or acetone will dissolve paint and cheap plastics. Any parts
made to run with gasoline will work with acetone just fine.
Besides we are using tiny amounts per gallon. Just a few ounces
per ten gallons of gas. Alcohol has been shown to be corrosive in
an engine yet they put THAT garbage into gasoline. Alcohol in
general is anti-mileage but the oil company stooges claim it is
wonderful. Most of the alcohol that enters your gas came here from
Europe as stale wine. The myth of renewable energy has been milked
by the Big Guys as much as possible. They know that alcohol is no
good in fuels. Look at what happened in Brazil. Millions of
engines and fuel systems were ruined in that country by alcohol.
But do the Big Guys want your engines to last? No, of course not.
They want you to buy new vehicles.
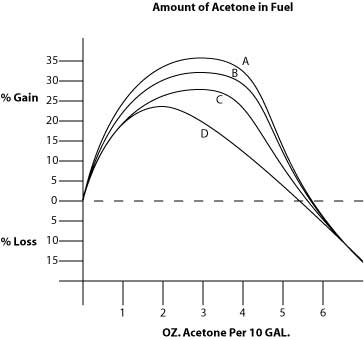
Shown is the percentage MILEAGE GAIN when a tiny amount of
acetone is added to fuel. The curves A B C
show the effect on three different cars using different
gasolines. Some engines respond better than others to acetone.
It is important to use the same gasoline from the same gas
station when testing mileage otherwise you have too many
variables and get undependable results. The D curve
is for diesel fuel. Too much acetone will decrease mileage
slightly due to adding too much octane
to the fuel. Too much also upsets the mixture ratio because
acetone (like alcohol) is a light molecule. Acetone helps the
fuel become a vapor more easily inside the chamber and mimimizes
wasted fuel.
After you find the
right amount for your car per ten gallons, and you are happy
with your newfound mileage, you may one day cease using acetone
for a couple of tanks. Watch the drop in mileage. It will amaze
you. That reverse technique is one of the biggest eye openers
concerning the use of acetone in fuel.
Complete vaporization
of normal fuel is far from perfect in today's cars. A certain
amount of fuel in most engines remains liquid in the hot chamber
and slides past the rings into the oil. Of course the liquid
fuel then ruins the rings and walls as it enters the crankcase
to further damage the lubricating ability of the oil. In order
to become a true gas and be fully combusted, fuel must undergo a
phase change. Still, fuel needs a kick of some kind to transform
from big globs into a full vapor. The acetone provides that kick
with its rapid inherent molecular vibration that prevents fuel
from escaping the combustion process and going through unburned.
In the chart above, we see how little acetone in ten gallons it
takes to help combust nearly all your fuel. Vacuum is the best
friend your engine has to get your fuel fully vaporized. At part
throttle the manifold vacuum reaches 20 to 25 inches of mercury.
This important vacuum aids in breaking up the larger fuel
particles and overcoming some of the disadvantages of surface
tension. Remember a slight amount of heat amplifies the surface
tension of the fuel molecules because the heat is spread across
the outside of the large clumps of fuel. This draws the clumps
tighter together. Just look how drops form. But a large amount
of heat will reach the inside molecules within the clumps. The
insides of the clumps must be broken up for complete combustion.
Most fuel molecules
are sluggish with respect to their natural frequency. For
instance the energy barrier from surface tension can sometimes
force water to reach 300 degrees before it vaporizes. Similarly
with gasoline. So any amount of water in the fuel is a killer to
proper combustion. Fuel is commonly forced to reach excessive
temperatures to vaporize. Your jump in mileage with acetone
comes from the (former excess) fuel that now gets burned and no
longer winds up in the crankcase. You stop the waste. That
excess fuel was formerly wasted past the rings or sent out the
tailpipe but now (with acetone) it gets burned. Conventional
fuels are not designed to fully vaporize or fully combust. This
simple fact appears to be deliberate on the part of the oil
companies although some companies such as Texaco, Chevron and
Canadian Shell deliver excellent gasoline mileage (in the
opinion of the author) and in view of repeated test results. On
numerous trips across the country in a 1995 Neon, his son
calculates the mileage and they get a consistent 49 MPG with
Texaco or Chevron. Often they get 40 in town. With a special FOG
Inducer device, they reached 52. A new
Bright Enterprises version exists that may raise the mileage
over 60 MPG in 2005. In fact we are testing numerous versions to
gain big mileage.
Many years ago the
author used a 1949 Olds to test fuels and immediately found that
100-percent acetone had way too much octane. About 150. So he
eventually wound up with a fuel mixture around half acetone and
half kerosene at the end of that experiment. By no means is this
any recommendation. Nor did he simply start mixing chemicals
without careful research and calculations. He knew enough about
organic chemistry to investigate fuels on paper prior to pouring
anything into a test tube or fuel tank. It was important to have
some reasonable prediction of the final results. Furthermore the
author modified his own carburetors, made carbs from scratch and
tailored MANY different fuel mixtures for test purposes. A
natural-born experimenter, he has been doing it ever since.
During extensive fuel
testing on an engine dyno in Wilmington, we evaluated the
effects of three additives on performance and economy. The three
additives sprayed into the intake were water, alcohol and
acetone. The spray entered just below the carburetor.
Performance and economy improved with each reduction of
the alcohol and/or water content of the spray mixture. But each
and every increase in the acetone content improved
performance and economy and reduced emissions. This correlation
was true up to a point because only a tiny amount of acetone
(fraction of one-percent) proved helpful. In other tests,
acetone in gasoline cut emissions by an honest 50-percent or
more. These tests were tedious but uncomplicated and verifiable.
It is a similar story
when testing diesel fuels and acetone. A tiny bit of acetone in
the fuel proved beneficial. The idle RPM goes up slightly by
about 150 and the truck gains power and torque. It also runs
more smoothly with much better economy. The difference is
obvious.
Alcohol was introduced
into our commercial fuels in recent years. But alcohol related
problems have systematically been kept from the public. A very
real cover-up has occurred for political reasons. Mileage drops
as high as 50-percent are real (in my own testing) for instance.
It make me angry. The biggest problem with alcohol in fuel is
due to surface tension. Surface tension (ST) is seen when you
place a drop of fuel, alcohol or water on a hot plate. It
collects into balls and dances around without vaporizing.
Ordinary fuel does not vaporize readily because of ST. Instead
fuel balls up and dances around on the hot plate. Surface
tension is like glue between fragments inside the fuel. So
within an engine, fuel (gasoline or diesel fuel) resists being
finely atomized due to ST. Complete atomization is essential to
good combustion but you are DENIED that with alcohol. Gasoline
needs injectors to produce a very fine spray into the air
stream. Unfortunately the sprayed fuel tends to recombine into
larger, persistent droplets because of surface tension. New
injectors usually improve combustion because of making a finer
spray pattern. With alcohol, gasoline and diesel fuel droplets
may fall out of the airstream onto the walls of the manifold.
Even in the most modern combustion chambers, some alcohol in
(mixed) fuels may not start to burn until the chamber
temperature reaches many hundreds of degrees. Good efficiency
and ST are diametrically opposed. Thus particle size remains
high when ST is prevalent. The real object should always be to
REDUCE particle size.
The best way to fight
many problems with fuels (such as ST) is to add just tiny
amounts of acetone to better operate your gas engines and/or
diesel engines. Acetone works wonders for engine Thermal
Efficiency. The author has used this additive since 1956 with
great success. The pure acetone label is the only additive
suggested and is easily available from most stores in 16-ounce
plastic bottles and in one-gallon containers from some large
farm supply stores. But any acetone source is better than none.
In a 10-gallon tank of gasoline, the author has generally used
one to four ounces of pure acetone to obtain excellent mileage
improvements. In a 20-gallon tank, the author has used about
four to eight ounces of acetone with the gasoline. In a
ten-gallon tank of diesel fuel, the author has used from 1 to 2
ounces of acetone and noticed that exhaust soot was greatly
reduced as fuel mileage rose significantly. Performance went up
too. A teaspoon of acetone in the fuel tank of a lawnmower or
snowblower is what the author normally uses.
The author suggests
not to continuously add acetone to each and every tank of fuel,
rather only to every other tankful. The slightest amount of
acetone seems to be highly effective for mileage and significant
emissions reduction. Most diesel fuel is oily and a tiny amount
of acetone has zero effect on injectors--in case that is a
worry. Any such problems would have shown up after nearly five
decades. In the past we have added the Torco two-cycle G7
smokeless synthetic gasoline additive to diesel fuel to make the
fuel even more slippery.
If you own a construction
or trucking company with heavy equipment and lots of
gasoline and diesel engines, how would you like to save roughly
30-percent on your fuel costs? How would you like your engines
to last twice as long? How would you like to get 15-percent more
power from your engines? We have nearly a half century of
experimentation with fuels and oils that you might find useful.
Our approach is simply to eliminate the waste that you normally
blow out the exhaust. That's it. Just stop the waste.
Ordinary gas or diesel
fuel allows a portion of the unburned fuel to bypass the
combustion process to escape in various forms of partial
combustion products and go out the blackened tailpipe. Some
unburned fuel enters the engine oil past the piston rings. The
incomplete combustion creates smog, pollution and poor economy
from wasted fuel. The surface tension of water is about three
times that of alcohol and most gasoline
components. A few drops of moisture in the gas causes the ST of
the fuel to zoom and not vaporize. Hence the partial products
cause wasteful results. Unfortunately many gas stations have too
high a tolerance for water in their tanks. It only takes a tiny
amount of water to ruin even the best gasoline. We need tighter
laws against water in stored fuel. Placing acetone in the
gasoline or diesel fuel helps greatly to break up the water and
alcohol the would otherwise cause any vehicle to run poorly and
waste fuel. There is an increase in water in the exhaust of an
engine running a tiny amount of acetone in the fuel due to
improved combustion. The more gas you successfully burn, the
more water you produce..
The acetone molecule
works physically to vigorously shake up every drop of fuel. It
acts like an internal vibrator to shake up each tiny bit of fuel
so the fuel does NOT ball up or glue together into large
aggregate particles. Instead this important additive guarantees
more complete vaporization of fuel inside the combustion chamber
where it really matters to defeat surface tension. Acetone
allows gasoline to behave more like the ideal automotive fuel
which is PROPANE. The degree of improved mileage depends on how
much unburned fuel you are presently wasting. Thus you might
gain 15 to 35-percent better economy from the use of acetone.
Sometimes even more. Winter is very hard on mileage. Cold
weather kills good mileage due to the reduction in Thermal
Efficiency. For this reason, the author only conducts outside
economy tests in the summer months.
Remember that acetone,
alcohol, gasoline and all fuels in general are extremely
flammable. Fuels in general should never be allowed near a flame
of spark source. Fuels can also dissolve asphalt, most plastics
and paint. They can damage skin and clothing. They should be
kept in properly ventilated storage and not mixed with unknown
substances. Keep children away from all dangerous chemicals. Do
not bring dangerous fuels inside your home. A garage is
typically a common place for such chemicals.
Why has the author
revealed the benefits of acetone in cars and trucks? To keep our
money in the U.S. even though the oil industry will do anything
to prevent better mileage. They will do anything to prevent
engines from lasting a long time. Why have they suppressed
acetone and propane for many decades? We must assume it is ON
PURPOSE. Simple GREED. Better mileage causes engines to last
longer due to less unburned fuel going past the rings, thus
keeping the rings and engine oil in far better condition. But
the biggest reason to conceal this additive from you is that bad
mileage gives the oil and car industries higher profits that
comes right from your pockets, ruins the air and throws our
children and our Nation into future debt. Do we want this
insanity and uncontrolled greed to continue? Tell your Senator
that we need acetone added to our gasoline.
Any person can easily
verify that acetone improves fuel mileage. This is a no-brainer.
Besides this is something YOU can do for your country. Send this
page to your Senator. A simple way to determine the way acetone
increases MPG is to document the number of days it takes going
to work before you need to refill. With the right amount of
acetone, you will go more days per tankful.
In conclusion, alcohol in fuel attracts water. This hurts
mileage because water acts like a fire extinguisher. It's not a
smart thing to put in cars or trucks. Some cars may run badly and
even quit due to the incombustible nature of the water laden fuel.
We know of a dozen cars that recently stopped running due to water
in the alcohol and gas mixture. They used the same Blue Planet
gasoline. (One of those was my 1986 Jimmy). The gas tank was
removed and drained. Look at the existing SUV situation with
horrible mileage. In cold weather (below zero), the water
and alcohol form nasty (abrasive) icy particles that can damage
fuel pumps. BUT acetone helps to fight the ice crystals. By adding
acetone for better mileage, fewer total emissions per mile get
blown into the atmosphere. Plus you can drive much farther. There
is less pollution, period. How sad that in the 21st Century, a new
30,000 dollar vehicle gets an unholy 12 to 17 miles per gallon.
Hey folks, my 1936 Dodge did better than that. What have the
(mighty and powerful) car and oil industries been doing since
1936? Not to mention the ineffective job the DOE is
doing--because it seems they are in bed together: DOE, car makers,
Big Oil, API and some members of Congress. And why have
conservationists such as the touted Sierra Club not recognized the
value of acetone in the combustion process? Why have they not
recognized that good mileage automatically means lower emissions?
What gives?
If John Wayne were
alive, he would probably tell you, "Hell yes, run acetone
in your car. Who do those SOBs think they are?"
We would like to hear
from car owners whose cars have quit after filling up with
gas-alcohol fuel. The author knows it is a trivial thing to
obtain great mileage. He has done it over and over for nearly 50
years by many different methods. The essential question is, "Why
won't THEY?"
I hereby swear all stated above is true and factual.