Research demonstrates that activated carbon could store hydrogen at room temperature
By Darren Quick
22:17 September 20, 2011
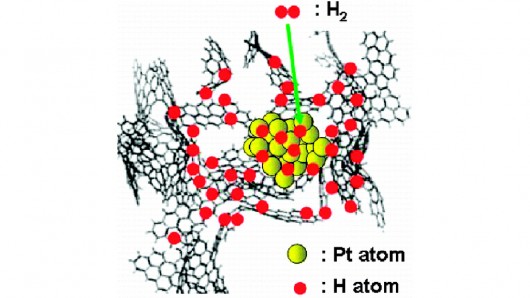
An MIT-led research team used inelastic neutron scattering to demonstrate that hydrogen atoms can diffuse to the carbon surface of platinum-doped activated carbon at room temperature
With hydrogen atoms consisting of just a single electron and single proton, its gaseous form made up of two hydrogen atoms can be hard to contain. Hydrogen storage, along with hydrogen production and the lack of infrastructure, remains a major stumbling block in efforts to usher in hydrogen as a replacement for hydrocarbon-based fuels in cars, trucks and even homes. But with the multiple advantages hydrogen offers, developing hydrogen storage solutions has been the focus of a great deal of research. Now an MIT-led research team has demonstrated a method that could allow hydrogen to be stored inexpensively at room temperature.
Hydrogen storage solutions fall into one of two technologies; physical storage where compressed hydrogen gas is stored under pressure or as a liquid; and chemical storage, where the hydrogen is bonded with another material to form a hydride and released through a chemical reaction.
Physical storage solutions are more established technologies but offer significant problems when looking at using hydrogen to fuel vehicles. Compressed hydrogen gas needs to be stored under high pressure - current hydrogen fuel cell vehicles such as the Mercedes-Benz F-Cell store hydrogen at 5,000 or 10,000 psi - which requires heavy tanks that add to the weight of a vehicle. Meanwhile, liquid hydrogen boils at -253°C (-423°F) so it needs to be stored cryogenically with heavy insulation and actually contains less hydrogen compared with the same volume of gasoline.
Although they allow hydrogen to be stored at much lower pressures, chemical storage solutions that bond hydrogen to a highly porous, sponge-like material such as a metal hydride generally require high temperatures to release their hydrogen content because most metal hydrides bind with hydrogen very strongly.
Previous research efforts have shown that a Rhodium-based material could store hydrogen at room temperature and would release the hydrogen when a small electric current was applied. Now an MIT-led research team has demonstrated that activated carbon can also store hydrogen under similar conditions and can release the hydrogen by simply releasing the pressure.
The activated carbon incorporates a platinum catalyst that allows the hydrogen atoms to bond directly to the surface of carbon particles and be released when needed. Because it allows hydrogen to be stored at atmospheric pressure and room temperature, storage tanks could be made much lighter, cheaper and safer, thereby making them practical and economically viable for hydrogen-powered cars, say the researchers.
To assess hydrogen's interaction with the activated carbon storage material, the research team used a technique called inelastic neutron scattering (INS). This method provided the first evidence that a phenomenon called the "spillover effect" was involved where, with platinum particles acting as a catalyst, hydrogen atoms split off from their molecules and diffuse through the carbon, where they bond with its surface.
Sow-Hsin Chen, MIT professor emeritus in the Department of Nuclear Science and Engineering and senior author of a paper describing the new method, says it should make it possible to increase the storage capacity of the activated carbon material by fine-tuning the size and concentrations of the particles of platinum and carbon. The team also hopes to identify a catalyst that isn't quite as expensive as platinum.
Once the storage system has been tuned to achieve the desired capacity, Chen says it should be capable of storing hydrogen under moderate pressure - possibly around 500 psi - and release the gas on demand by simply releasing the pressure. This is because when the hydrogen molecules are broken down into atoms using the spillover effect, they bind with the activated carbon with much less energy.
Chen led the research team, which included former MIT student Yun Liu, now at the National Institute of Standards and Technology and the University of Delaware; and researchers at Taiwan's Institute of Nuclear Energy Research (including lead author Cheng-Si Tsao, who was a visiting scientist at MIT for a year working with Chen), National Tsinghua University in Taiwan and Pennsylvania State University.
Their findings were reported in a paper published online in the Journal of Physical Chemistry Letters in August and is scheduled to appear in an upcoming print issue.
Source: MIT news
Copyright © gizmag 2003 - 2011 To subscribe or visit go to: http://www.gizmag.com